|
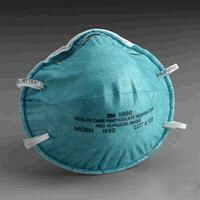
Click to enlargepad3Mâ„¢ 1860 Health Care N95 3Mâ„¢ 1860 Health Care N95 Particulate Respirator and Surgical Mask. NIOSH approved as a Type N95 respirator. It meets CDC guidelines for TB exposure control and is designed specifically for use in a health care setting. Recommended by health care officials as precautionary measure against the "bird flu" and SARS. It has a soft inner shell for greater comfort against the face and, because it's fluid resistant, it aids in reducing the potential exposure of the wearer to blood and other fluids. * NIOSH approved as a Type N95 respirator to meet OSHA requirements and CDC guidelines for TB exposure control. * Helps reduce wearer exposure to airborne particles. * >99% BFE1 to help reduce patient contamination caused by exhaled microorganisms. * Fluid resistant to help reduce potential exposure of the wearer to the spray, spatter and aerosol of blood and body fluids. * Does not contain either natural rubber latex or dry natural rubber as components in the product or its packaging. * Respirator includes a soft inner shell for greater comfort against the face. * Hypoallergenic to reduce chance of allergic reaction. * 1 - Bacterial Filtration Efficiency (BFE) determined by the modified Greene and Vesley test method. BP Medical Supplies Brooklyn , NY 11236 (***)-276-2236 (3M 1860 N95 safety respirator surgical mask 20/bx is the responsibility of Marcia Hughes) |
m_hughes@cedar--rapids.com (Marcia Hughes) for more information.